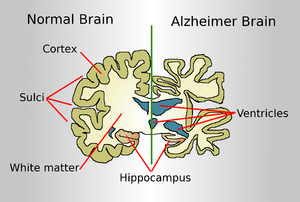
Alzheimer’s disease is a progressive, chronic disease that attacks the brain cells and neurons; dementia is the first clinical symptom of this disease, which normally afflicts people in the old age. Alzheimer’s is a serious illness that impairs the cognitive skills of the patient to such an extent that they need to be under the supervision of assisted care. Such an ailment could be understood better if scientists identify the metals of the brain, which are involved in instigating the neurodegenerative condition. Such a research study was recently completed by an international group of researchers through a collaborative approach. The research team was headed by Dr. Joanna Collingwood, who works at the School of Engineering, University of Warwick.
This research team reported that iron species in the brain could be characterized easily as they play a pivotal role in the formation of plaques caused by amyloid proteins; these proteins are extensively found in the human brain. Clustering of these proteins may reach an abnormal level, leading to the formation of plaques. The toxicity of these plaques is severe, causing apoptosis (programmed cell death) of tissues. With the progressive of tissues in the brain, the person’s cognitive skills decline consistently; memory loss is followed by deterioration in mental condition and the patient ultimately develops Alzheimer’s disease.
In patients with Alzheimer’s disease, the plaques formed by amyloid proteins are unique and can be characterized easily. The iron species are otherwise in their normal state in a healthy human brain. When a person develops Alzheimer’s disease, these iron species undergo extensive chemical reduction. This leads to the formation of a chemically reduced species named magnetite; the proliferation of magnetite occurs in the amyloid protein plaques. The team of researchers believes that magnetite is the result of the chemical interaction between iron species and amyloid proteins in the human brain afflicted with Alzheimer’s illness.
In advanced countries, like the USA and UK, there have been several advancements in diagnostic technologies used for the analysis of human brain. One such advancement is the introduction of Diamond Light Source 108 beamline instrument in Oxfordshire, UK. This sophisticated instrument is capable of performing advanced measurements of the human brain using synchrotron X-rays. The team of researchers at the University of Warwick used this instrument to gather evidences for their work. They were successful in proving that detailed chemical reduction of metallic species only took place in the human brain of individuals afflicted with Alzheimer’s disease. They further reported about the different forms of calcium species that were present in the form of minerals in the plaques formed by amyloid protein.
What exactly is the significance of this discovery? Well, we have now understood that metals in the human brain undergo a lot of chemical reduction and transformation when a person is afflicted with Alzheimer’s disease. This would help us in further discovering the root cause of the disease. By correlating the concentration of these reduced metal species with the progression of the disease, scientists can develop more innovative therapies that can exactly eradicate the root cause of the disease.
Dr. Joanna Collingwood, who is the supervisor leading this team of international researchers, works as an Associate Professor at the School Engineering, University of Warwick. She is an expert in following analytical techniques of measurement: trace metal analysis and high resolution imaging. She has used these skills to understand and elucidate the pathophysiology of neurodegenerative diseases, including Alzheimer’s disease.
According to Dr. Collingwood, we need to know the guiding principle of this research study: Iron is an essential element in the human brain, so it is but natural that we comprehensively understand how the fluctuations in iron levels could be associated with the development of Alzheimer’s disease. It is important to note that we are only concerned about iron levels in the human brain at this stage of research. We used sophisticated X-ray techniques in this study to understand how iron undergoes a step-wise change in its neurochemistry while interacting with amyloid proteins, which cluster together excessively to form plaques. It is interesting to note how amyloid protein formation instigates a massive chemical change in the composition of iron species, which are found extensively in the human brain. Previous studies have attempted to treat Alzheimer’s disease with iron-based drugs. Our findings would only alleviate further research studies in this direction, leading to the development of novel drugs based on iron.
The research study was conducted by an international collaboration between researchers working at the following institutions: University of Warwick and Keele University. It also included researchers working at the following institutions: University of Texas in San Antonio and University of Florida. To form an insight into this path-breaking discovery, the team of researchers first successfully extracted the cores formed by amyloid protein plaques; these were obtained from the human brain of two deceased patients who had succumbed to Alzheimer’s disease.
For the purpose of scanning the cores formed by amyloid plaques, researchers used the following sophisticated analytical instrument: an advanced X-ray microscope was obtained from the Advanced Light Source in Berkeley, USA. The Diamond Light Source synchrotron (beamline 108) was also used in this study; this instrument was obtained from Oxfordshire and its main purpose was to determine the chemical properties of the altered minerals found in the brain of these deceased patients. With the help of these sophisticated instruments, the researchers also analyzed whether all the iron species had undergone a change in its magnetic state when they interacted with the amyloid proteins in the plaques. Researchers tried to find out if there were any unchanged iron species in the plaques. Magnetite, the leading magnetic form of iron oxide, was found extensively in these plaques. Other minerals of altered iron species were also found in these plaques.
Significance: The UK is a small country with the best healthcare system in the world. Sadly, it still cannot cure about 850,000 patients diagnosed with dementia, the first clinical symptom of Alzheimer’s disease. The number of patients with Alzheimer’s disease is expected to be skyrocketing at 1 million and 2 million by 2025 and 2050, respectively. Presently, medical science has no cure for this disease and other conditions that cause dementia. If the development of dementia in Alzheimer’s patients is delayed by at least five years, the number of deaths caused by this condition could be halved easily. This implies that approximately 30,000 patients could be prevented from untimely death; their lives can be saved with this path-breaking discovery.
Conclusion: Researchers believe that the development and progression of Alzheimer’s disease is associated with the chemical reduction of iron species in the human brain. The resultant iron species have an altered magnetic state; the most prominent among them being magnetite. All these altered iron species are highly toxic in nature, creating conducive conditions for Alzheimer’s disease.