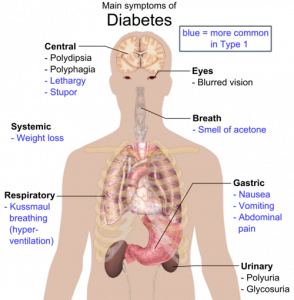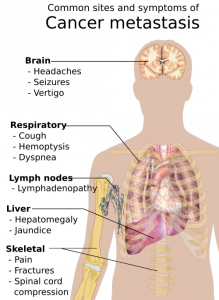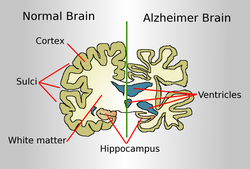
Doctors have to refer to randomized clinical trials to determine the best ways of treating patients. Furthermore, doctors refer to this information to also determine the most suitable drug that can be prescribed to these patients. Heart illness is the most common disease that afflicts the common man. In recent years, it has been proven that the number of women having heart ailments would be greater than the number of men having the same ailment. Compared to younger people, older people have a greater tendency of developing heart condition. Does the data presented in clinical trials actually exhibit reality?
In most cases, the data does not actually represent the true picture. A new study was recently published in the journal Circulation: Cardiovascular Quality and Outcomes. Professor Quoc Dinh Nguyen works at Université de Montreal’s Faculty of Medicine. He supervised a team of researchers who tested new heart drugs on mostly men (71 per cent); however, the majority of people afflicted with heart disease were mostly women. Moreover, the average age of male patients with heart disease was 63; however, the average age of patients who suffered the two most common heart diseases was in the range of 68–69 years.
In the past 20 years, the gender and age gap between subjects participating in drug trials has hardly diminished; however, the population seems to be aging rapidly. Professor Quoc Dinh Nguyen is a geriatrician who works at the Centre Hospitalier de l’Université de Montréal (University of Montreal Hospital Center).
In most drug clinical trials, both women and older patients are under-represented; therefore, both these groups of patients would receive comparatively lesser care. Unlike a young patient, an older patient does not really respond much to several treatments and medications. Many-a-times, it is very difficult for patients to receive an exact dosage or intervention; moreover, each medication has a set of severe side-effects. However, we do not know the specific course of medication until and unless a large number of older patients are included in clinical trials. Most studies also do not include women in their clinical trials.
In the present scenario, the findings are obtained from a clinical trials conducted predominantly on male and younger subjects. These results do not really patient outcome of women and older subjects generally. Nguyen examined the issue closely while working as a geriatric resident. They discussed with colleagues to realize that heart disease is a grey area to receive effective treatment. Resident physicians of other departments (anaesthesiology, psychiatry, emergency medicine, and cardiology) also collaborated in their efforts of improving the results.
A brief history
Nearly 20 years ago, researchers were concerned about how under-represented were several sections of the society, especially women. The results of clinical trials were quite often problematic in nature. With a team of researchers headed by Nguyen, we set off to find out if these practices had improved significantly. The 25 most frequently cited clinical trials were examined closely every year. The examination period was of twenty years, ranging from 1996 to 2015. The data was published in the U.S National Health and Nutrition Examination Survey 2015-2016; this data compared how prevalent was cardiovascular disease in America . The data was classified in terms of following parameters: age and gender.
The research team examined data of following medical conditions: coronary artery disease, hypertension, heart failure, atrial fibrillation. This team also examined several risk factors contributing to cardiovascular diseases. This research team closely examined the correlation between diabetes and heart disease. Previous studies have reported that diabetic patients were more likely to suffer from coronary heart disease.
Bad results
Currently, a greater number of women and older patients are included in clinical trials; therefore, there has been a slight improvement in the representational bias of clinical trials. Eric Peters works as an anesthesiologist at the CHU Saint-Justine children’s hospital; he is the second author of this study. Depending on our calculations, it would take another 90 years to understand whether clinical trial studies could present data correctly without bias. We need to correctly understand the factors contributing to coronary heart disease. The factors leading to aging population must also be considered in this situation.
After analyzing 500 clinical trials, we arrived at the following conclusion: only 29 percent of participants were women in this clinical trial. Moreover, the average age of participants was just 63 years. According to Nquyen, the reality is quite different in the hospital emergency rooms and departments; these departments are of internal medicine, cardiology, and geriatric medicine. Women and older patients were hardly represented in clinical trials that were focused on determining the factors associated with CAD and heart failure. Women represent more than 54.6 percent of CAD patients. In clinical trials, more than 27.4 percent of participants for CAD were women.
Is heart disease really a man thing?
It is a general perception that men are afflicted with heart disease; however, most medical research studies have reported about results that are completely obsolete. Heart disease is the leading cause of women’s death in Canada. Fewer men die of heart disease in Canada. In general, heart disease would affect women at a later stage in life. Nearly after 10 years, women would die of coronary artery disease and heart failure. There has been steady decline in the death caused by heart disease in women; moreover, death caused due to heart disease would be greater in men. One of the most common hypothesis is the fact that men receive timely medical treatment unlike women.
Why are women excluded from clinical trial?
In general, most women were excluded from clinical trials because it was advisable to give them medication during pregnancy; however, this principle should not have been applicable to drugs used for treating heart condition. This is because most patients with heart conditions are usually more than 60 years of age. To select a woman to participate in clinical trial, we also need to consider the age of the woman. To ensure the adequate participation of women in clinical trials, older patients should be recruited. This is because women are afflicted with cardiovascular disease at a comparatively later stage of treatment, unlike men.
In general, it is difficult to conduct clinical trial of older patients. This is because most older subjects would find it difficult to move around; moreover, it is usually tougher for them to undergo a battery of clinical tests. Older the patients, higher would be their difficulty in moving around. In general, older patients do require several medications as they are afflicted with several ailments.