To promote the functioning of beta cells and insulin in patients with type 1 diabetes, researchers have developed a novel strategy that minimizes the requirements of insulin and the incidence of hypoglycemia. These researchers have worked at the Comprehensive Diabetes Center at the University of Alabama in Birmingham, USA.
The journal Nature Medicine published these findings recently. Verapamil is the most commonly used drug for controlling blood pressure; it was approved for oral administration in the year 1981. Following the administration of verapamil, type 1 diabetes patients were able to produce greater amounts of insulin. Thus, their daily requirement of insulin was reduced substantially and their blood sugar levels were also in control. The drug verapamil is not just safe and effective for type 1 diabetes but also a promising therapy that provides new hope to people living with this life-threatening condition. These were positive results confirmed in a human clinical trial that was randomized and double-blinded in nature; the clinical trial was controlled by placebo.
Verapamil has shown promising results in improving the function of beta cells in pancreas; the functioning of beta cells is related to the control of insulin production. Optimum levels of insulin ensure a good quality of life in patients. Under such a scenario, type 1 diabetes patients have new hope. It is otherwise difficult to control such a life-threatening condition that offers no hope. Although this drug does not sound like a complete cure for type 1 diabetes, it is however a promising therapy for altering a life-threatening condition: type 1 diabetes. Such patients need to boost the production of insulin in their body in order to have better disease control.
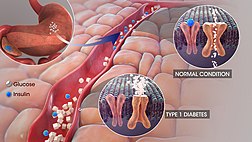
A clinical trial was conducted on animal models in the year 2014. In this clinical trial, it was reported that the condition of type 1 diabetes could be completely reversed by administering verapamil. Then, they conducted a human clinical trial to determine the effects of this drug. For more than three decades, the drug verapamil has been approved by the FDA for the treatment of high blood pressure. Current research findings are path-breaking in the sense that the drug is quite safe and effective in the treatment of type 1 diabetes patients. In patients with type 1 diabetes, the body’s immune system attacks beta cells of the pancreas. These beta cells are responsible for the production of insulin. Insulin is the hormone that controls blood sugar levels in the patient.
The production of insulin decreases substantially when the beta cells of pancreas are destroyed in the human body. Consequently, blood sugar levels would rise in the human body and the patient would become extremely dependent on external sources for insulin. The function of beta cells can be preserved effectively when a patient is administered verapamil. This drug induces the body to produce more insulin. In various clinical trials, it has been proved that the participants’ dependency on external insulin decreases substantially. Several individuals with type 1 diabetes can effectively regulate their blood sugar levels with this strategy.
In the human clinical trial, the drug verapamil was administered to 24 patients. These patients were in the age group of 18 to 45 years. Over the course of one year, verapamil was administered to 11 patients while a placebo drug was administered to 13 patients. Only patients with type 1 diabetes were included in this clinical trial. They received insulin therapy to manage their condition throughout the duration of this clinical trial. The total daily dose of insulin was monitored in both the groups, that is, the group that received verapamil and the group that received placebo. Moreover, we also monitored the amount of insulin produced in these groups. Factors such as the percentage of change in insulin and HbA1C levels were also monitored. There were patients who experienced hypoglycemic events; all such events of each patient were recorded in our human clinical trial. A continuous glucose monitoring system was used to determine the healthy blood glucose levels of each patient.
Patients with type 1 diabetes do have therapeutic options for hope. In fact, such patients should be able to deal with the illness in a promising way following the successful administration of verapamil. Insulin dependency was substantially reduced in patients with type 1 diabetes following the administration of verapamil. The quality of life was significantly improved in these patients. The risk of comorbidities would be improved when the overall blood sugar levels was controlled in patients. Thus, a patient with type 1 diabetes would not develop several other comorbidities, such as kidney disease, blindness, and heart attack.