The University of Pennsylvania has conducted a promising clinical trial to devise a new type of vaccine for cancer. Although the clinical trial was of an initial stage, promising results have been meted out and researchers are hopeful of a breakthrough discovery.
The clinical trial was a joint collaboration between researchers of following medical schools, which are affiliated to the University of Pennsylvania: the Perelman School of Medicine and the Abramson Cancer Center. The vaccine is truly innovative in the sense that it corporates the immune cells of patients; the immune cells are directly exposed to the tumor cells of patients.
This experiment was carried out in a laboratory under simulated conditions. Following treatment, the immune cells are then injected into the patient to elicit a better immune response.This experimental clinical trial was performed on patients diagnosed with ovarian cancer at an advanced stage.
This was a pilot trial whose sole purpose was to determine the feasibility and safety of novel vaccine; however, the results were promising enough to ensure that is very effective in nature. Anti-tumor T-cell responses were elicited in more than half of the patients that participated in this clinical trial.
Patients that responded to this treatment had higher life expectancy despite tumor progression unlike patients who just did not elicit any response. In fact, one patient became “disease free” for five years after receiving being treated with this vaccine for two years. The promising results of this clinical trial have been published currently in the journal Science Translation Medicine.
The lead author of this study was Dr. Janos L. Tanyi, MD, who works as an assistant professor of obstetrics and gynecology at Penn Medicine. The researchers concluded that the novel vaccine was safe for clinical treatment of patients. This vaccine elicited a broad anti-tumor immunity; however, they have strongly recommended more clinical trials on a larger scale.
The other researchers who worked with the lead author at the Perelman School of Medicine at the University of Pennsylvania are as follows: Lana Kandalaft, PharmD, PhD, George Coukos, MD, PhD, and Alexandre Harari, PhD. The conventional treatment offered by cancer vaccines can be summarized as follows: A cell-surface receptor is a specific molecule that is mostly attacked by most cancer vaccines till date.
This molecule is generally found on cancerous cells in any kind of tumor. However, the team headed by Lausanne-Penn devised a far more aggressive approach. They developed a personalized vaccine that took into considered every individual cancer patients’ condition. For this purpose, they comprehensively analyzed the tumor system of each cancer patient.
The set of mutations are unique to each tumor, presenting a unique pathology of the impaired immune system. With this information, they developed a whole-tumor vaccine that elicited immune response and combated not just a single target in the tumor, but about hundreds or thousands. This is a truly innovative strategy that outshines the efficacy of conventional vaccines.
The basic objective of this clinical trial was to elicit a strong immune response that targets tumors comprehensively. They were successful in eliciting an immune response that hits all kinds of markers, including the markers that are unique to a particular tumor.
The formidable defenses of tumors were overcome by harnessing the T-cell immunity with the vaccine. To prepare a personalized vaccine for each patient, the researchers sifted through the mononuclear cells of peripheral blood, which was obtained from each patient.
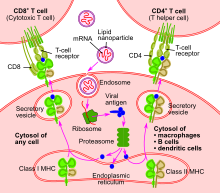
They identified precursor cells that were suitable enough for use in this clinical experiment. These cells were grown into a culture in a laboratory under carefully controlled conditions. Thus, they produced a large number of dendritic cells. A T-cell immune response can be effectively elicited with the use of dendritic cells.
Infectious pathogens are engulfed by these T-cells; moreover, these T-cells also engulf tumor cells and anything that is considered “foreign”. Nevertheless, a specific response is elicited by the patients’ immune system when T-cells and other components of the immune system are again exposed to pieces of invader cells.
The patients’ tumor cells were obtained and a special extract was prepared from these tumor cells. Then, the extract of tumor cells was exposed to dendritic cells; the dendritic cells were activated by irradiating them with interferon gamma. Finally, the patients’ lymph nodes were injected with these activated dendritic cells and a T-cell response was generated.
The team of researchers successfully carried out this strategy on 25 patients in total. Every three weeks, each patient was administered a dose of dendritic cells; it is important to note that these dendritic cells were treated with tumor cells by a process described above.
The exposure of dendritic cells at periodic intervals was carried out for six months. A huge increase in the number of T-cells was reported in more than half of the patients included in this trial. What’s more fascinating is the fact that generated T-cells were specifically reactive to tumor cells. In other words, the personalized vaccine developed for combating cancer was hugely successful.
The patients that responded to this treatment showed 100 percent survival for a period of two years. The patients that failed to respond to this treatment showed an overall survival rate of just 25 percent over a two-year period.
In this experiment, researchers had a included a stage 4 ovarian cancer patient who was 46 years old. The prognosis of this patient is generally very poor with conventional treatment, which includes five courses of chemotherapy. Interestingly, this patient remained disease-free for five long years after receiving 28 doses of the personalized vaccine over a two year period.
In conclusion, the researchers hope that the efficacy of this personalized vaccine would be doubled if it is combined with chemotherapeutic drugs that strive to suppress anti-immune responses of the tumor.